Applied mechanical processes stimulate the binding force between surface molecules required for parylene adhesion to substrates, which is essential to both good parylene performance and assembly/component functionality.
Basic Thermal Properties of Parylene Conformal Coatings
CVD-generated parylene combines high thermal stability with a low dielectric constant, minimal moisture absorption, and other advantageous properties which sustain its adhesion to substrate surfaces. Among the most beneficial of the parylenes’ thermal properties is their ability to function at an exceptional range of temperatures. Depending on the parylene type, they are operative at temperatures as low as -271° C, and as high as 450° C, representing an ability to perform within a span of 721° C.
 Much depends upon the specific parylene type, its explicit product purpose, and the environmental conditions affecting performance. However, when parylene type and purpose are appropriately matched to the expected thermal conditions of the assembly’s operational environment, parylene conformal coatings offer superior adhesion and minimal delamination.
Much depends upon the specific parylene type, its explicit product purpose, and the environmental conditions affecting performance. However, when parylene type and purpose are appropriately matched to the expected thermal conditions of the assembly’s operational environment, parylene conformal coatings offer superior adhesion and minimal delamination.
For instance, Parylene C can endure constant exposure to 100° C for eleven+ years, accounting for 100,000 hours of use, without appreciable delamination. In contrast, more recently developed parylene HT is useful in high temperature applications (short-term up to 450°C), although this represents an extreme range. More generally, the parylenes can provide similar service (11.4 years of persistent adhesion) in vacuums or atmospheres free of oxygen, working through ongoing exposure to 220° C, making them an excellent choice as conformal coatings for aeronautics’ and space flight uses. Higher temperatures can shorten parylene use-life in oxygen rich environs.
For most terrestrial uses, heat-treating for three hours at temperatures of 140°C, beneficially activates longer-term adhesion and insulation. Parylene’s low thermal expansion helps it retain uniform conformal qualities through innumerable functional settings.
Further thermal properties of parylene types C, N and D are provided in the table below:
| Properties | Parylene C | Parylene D | Parylene N |
|---|---|---|---|
| Melting point | 290°C | 380°C | 420°C |
| T5 point (where modulus = Taken from secant modulus temperature curve) | 125°C | 125°C | 160°C |
| T4 point (where modulus = Taken from secant modulus temperature curve) | 240°C | 240°C | 300°C |
| Thermal conductivity, 25°C | 2.0°C | -- | 3.0°C |
| Specific heat, 25°C | 0.17°C | -- | 0.20°C |
Optional Methods of Heat Pre-treatment
Heat treatments to improve parylene for adhesion may occasionally include:
Parylene coatings exhibit dependable consistency for many applications where exposure to ongoing thermal pressure is the rule. However, in some circumstances parylene films covering component substrates become fragile and inflexible due to persistent thermal stress, reducing their usefulness as conformal coatings. Unfortunately, cases of diminished coating adhesion have particularly presented themselves for biomedical implant applications, where malfunction may be life threatening. Improving these performance conditions is a significant challenge to the development of more adaptable parylene conformal coatings.
Parylene may be annealed to increase cut-through resistance, enhance coating hardness, and improve abrasion resistance. This is the result of a density and crystallinity increase, occurring after contact with heat.
At the same time, properties of crystallinity and surface morphology generally undergo some degree of transformation during deposition and thermal annealing, affecting parylene film adhesion, as well. These conditions suggest that, with proper treatment, conformal coating properties can be adapted to specified production details. Thus, the incidence of failure due to film delamination can be limited, if processes are carefully and thoroughly implemented, according to conditions of projected product use. Typically, substrates are pretreated with A-174 silane.
However, the fact that thermal stress generated in the film during CVD processing may weaken the conformal coatings’ adhesive force may further oblige customized coating procedures, to ensure delamination or similar adhesion failure does not become an issue. For instance, in certain cases, excessive temperatures (+ 150°C), and prolonged exposure to thermal sources – longer than 20 minutes – can lead to coating degradation, particularly if the films’ thicknesses are <3-μm and exposed to the higher temperature ranges. Degraded adhesion following annealing at 150°C for 20+ minutes can impair parylene’s encapsulation properties. While heat treatments of even 80°C may initiate thermal stress -- somewhat compromising the parylene conformal coating, leakage rates’ prevention -- factors of diminished adhesion generally do not significantly degrade the film at 150°C for the longer service durations (11.4 years), suggested previously.
Therefore, despite some inconsistencies of performance under conditions of thermal stress, parylene’s properties generally provide good thermal adhesion and endurance, in comparison to competing coating materials, for innumerable products and purposes. In some cases of terrestrial application, higher operating temperatures may shorten parylene’s functional life; oxygen-free, space vacuums are not affected by these conditions and can operate for similar timeframes at higher temperatures (220°+ C). Testing the complete structure conformally coated by parylene under conditions closely resembling intended operating settings is recommended to verify its ability to withstand or exceed these temperature-time-atmospheric conditions.
However, parylene reliably performs under most situations -- in air or a vacuum – at a wider temperature range than competing conformal coatings. It does so for a decade or more, at an extreme range of temperatures, without significant loss of physical properties, providing superior adhesion and limited delamination.
The Need for Adhesion Testing
Applied mechanical processes stimulate the binding force between surface molecules required for parylene adhesion to substrates, which is essential to both good parylene performance and assembly/component functionality. The emergence of conditions characterized by non-adherence and delamination squander parylene’s typically exceptional substrate protection against chemical attack, corrosion and moisture, as well as its superior dielectric insulation (er = 3.1).
 Parylene adheres poorly to low surface energy substrates, under any circumstances, leading to delamination, separating from the surface its meant to protect. A variety of preparatory substances -- including hexane, propylene carbonate (PC.), A-174 silane, tetrafluoromethane (CF4) plasmarface, and toluene -- can significantly improve interface adhesion between the substrate and parylene, when applied to the substrate surface prior to CVD.
Parylene adheres poorly to low surface energy substrates, under any circumstances, leading to delamination, separating from the surface its meant to protect. A variety of preparatory substances -- including hexane, propylene carbonate (PC.), A-174 silane, tetrafluoromethane (CF4) plasmarface, and toluene -- can significantly improve interface adhesion between the substrate and parylene, when applied to the substrate surface prior to CVD.
Suitable Methods of Parylene Adhesion Force Testing
Improving parylene adhesion requires a close review of all of the current processes affecting its bond with the selected substrate. As with all aspects of the adhesion process, care needs to be taken in this regard. At present, there is no single widely-accepted test for conformal coating adhesion. Determination of adhesion priorities needs to be made on a case-by-case basis. It is very important to match the test methodology to the type of parylene being used, and the expected conditions under which the component will function, when operative.
ASTM D3359 – Tape Testing for Adhesion
The Tape Test consists of two variations:
ASTM D3359 is valuable because it offers a high standard for adhesion testing; passage verifies exceptional levels of conformal performance for parylene bonding to the selected substrate. Tape testing is very similar to the "Knife Test" described below.
Knife, Pull-off, and Scrape Tests
Additional testing methods of value for verifying parylene adhesion include:
Whatever method is used, it is a mistake to opt for too-rigorous an adhesion standard for parylene films. Parylene has repeatedly demonstrated its capacity to provide superior conformal coating under exceptionally ruggedized conditions. While its adhesion capacities should always be subjected to appropriate testing, there is no value in creating test standards so demanding that a new collection of coating materials must be invented to meet them.
Conclusion
Inadequate adherence of parylene conformal coatings significantly lowers the operational life of components and parts, causing them to malfunction during use. This can be a problem because the chemical structures of the parylenes may actually reduce good interface adhesion, sometimes significantly. Systematic cleansing of substrate surfaces stimulates better interface adhesion.
Heat-treated parylene (140°C, 3 hours) can also respond beneficially, engendering dependable adhesion and film insulation, minimizing delamination and other coating erosion. Heat testing can determine whether a high, consistent adhesion strength is the outcome, as it generally is with heated parylene. Appropriate heat-treatment can generate parylene adhesion-improvement as much 800% greater for conditions of prolonged operational duration.
In comparison to using parylene alone, bilayer encapsulation of PCBs and related assemblies improves the overall adhesion and performance-stability of active, wired devices, particularly in cases where superior adhesion is essential to persistent functionality.
Characteristics of Noble Metals
Selecting the appropriate pre-treatment procedures is a key factor to this success of parylene adhesion to any substance. Procedures vary quite considerably, according to the materials designated for conformal coating and substrate. Chemically inert surfaces like gold, silver and other noble metals, and nonpolar thermoplastics such as parylene, are extremely difficult to bond; they require additional surface treatments besides cleaning.
Managing noble metal adhesion is further complicated because parylene sticks to itself, rather than the substrate surface, raising difficulties for suitable attachment on the smooth surfaces typical of assembly components formulated of noble metals. Thus, in usage where the substrate bond is subjected to sliding friction or comparable forces applied perpendicular to the surface, the parylene coating may break away. Similar adhesion difficulties emerge for parylene concerning most functional applications where it is applied as a conformal coating for noble metal substrates.
 This is less the case where a metal substrate has a high RA, the arithmetic average of a particular substrate’s roughness parameters; higher RA signifies a sufficient quantity of surface cavities (flaws and fissures) to capture parylene, holding it to the surface, prompting acceptable levels of adhesion and diminished tendency toward delamination. However, once refined, the RA of most noble metal surfaces is low. The minimal micro-porosity of these surfaces is absent the required quantity fissures or flaws for generating longer-term parylene adhesion, mandating application of adhesion promotion techniques.
This is less the case where a metal substrate has a high RA, the arithmetic average of a particular substrate’s roughness parameters; higher RA signifies a sufficient quantity of surface cavities (flaws and fissures) to capture parylene, holding it to the surface, prompting acceptable levels of adhesion and diminished tendency toward delamination. However, once refined, the RA of most noble metal surfaces is low. The minimal micro-porosity of these surfaces is absent the required quantity fissures or flaws for generating longer-term parylene adhesion, mandating application of adhesion promotion techniques.
Improving Surface Energy
Without appropriate treatment, fundamental limitations inherent to parylene can render it unsuitable for applications exposed to friction, pressure, heat or thermal cycling. Parylene is inert, very soft and exhibits poor “creep” attributes during untreated application to noble metals. A general absence of active sites for forming intra-molecular bonds results in poor adhesion, a propensity to dislodge from the substrate; delamination and similar non-adhesion problems can occur if the surface is unmodified prior to CVD.
The importance of cleanliness-processing and masking pre-CVD to improve parylene adhesion have already been discussed, as has the use of Methacryloxypropyltrimethoxysilane. This A-174 silane compound is the surface treatment of choice for most applications where developing a reliable, longer lasting adhesive bond between parylene and a noble metal substrate is desired. Adhering chemically to the metal, the introduction of A-174 silane provides precisely the kind of uneven, flawed surface that stimulates parylene attachment, helping it to bond far more conformally to the generated surface cavities and fissures during CVD.
Application of A-174 is implemented through soaking, spraying or vapor phase techniques. Spraying is recommended when only selected portions of the substrate require treatment; soaking or vapor phase are suggested modalities for treating an entire assembly or component. Appropriate operational caution needs to be observed during processing of A-174 silane adhesion promoter; it is a moderate skin, respiratory, and eye irritant. Although not overly flammable, it is combustible, and should be handled with care. While A-174 silane is favorably endorsed throughout the industry as a surface treatment for noble metals requiring the benefits of parylene conformal coating, other treatment methods offer advantages of their own.
Surface Treatment Alternatives to A-174 Silane
Plasma polymerization processing can improve the adhesion and barrier properties of conformal films deposited on such noble metals as platinum; systematic approaches tailored to specific coating requirements are suggested. This approach has been successful as a pre-treatment for MEMs and nano- applications. Recommended for use in aggressive, harsh biomedical environments, plasma activation technology positively energizes the surfaces of many noble metal substrates, and is implemented immediately prior to CVD processing. In this regard, recent research demonstrated chemical oxygen plasma insertion pretreatments characterized by microscopic and surface-sensitive techniques could increase barrier hydrophilicity and surface energy for metal implant coatings, improving their overall biocompatibility and performance. This evidence suggests further development of parylene coating functionality, based on applications of plasma surface treatments for parylene coatings intended for noble metal substrates.
Mechanical abrasion processes treat noble metal surfaces with a very fine industrial grit. The objective is roughening the smooth surfaces by scraping them with the grit through tumbling or similar industrial processes, lightly abrading the designated substrates. Parylene coated wire mandrels used in manufacturing biocompatible metal tubing for medical devices can be pre-CVD treated with abrasion processes before parylene application.
Nevertheless, chemical bonding pre-treatment with A-174 -- applied either through soaking, spraying or vapor deposition -- remains the most commonly used pre-treatment to promote parylene adhesion with noble metals. The consequent chemical surface-bond is significantly improved by this mechanically-induced method. Where pretreatment procedures are appropriately implemented, they forestall coating delamination and enhance the effectiveness of conformal parylene corrosive barriers for noble metal substrates and devices.
Chemical vs. Mechanical
Parylene only adheres to substrates mechanically, and this can require assistance from additive substances; parylene’s chemically-based adherence is nonexistent. Adhesion is a consequence of molecular attraction stimulating the surface unification of two dissimilar substances; their joining creates a significant physical bond between them. Of the two primary types of adhesion, chemical adhesion results when a compound joins with another, because they share sufficient mutual chemical interaction to form a bond with each other. Because parylene is chemically inert, chemical adhesion is impossible; it adheres using the other method -- mechanical adhesion. Applied mechanical processes can stimulate this binding force between surface molecules.
One might think chemical substances are suitable to stimulate superior adhesion of parylene coatings, since its substrate-application employs the unique chemical vapor deposition (CVD) process. However, this is not so. In fact, the opposite is true. On their own, the chemical structures of virtually all parylene types (C, D, N, etc.) undercut good interface adhesion. The chemical vapor deposition (CVD) process that generates so many of parylene's benefits also nullifies chemically-based substrate adhesion; only mechanical adhesion is possible.
 For instance, low surface energy materials, such as Kapton (polymide), limit parylene for even mechanical applications, and entirely reject chemical adhesion, causing rapid delamination and peeling of the conformal coating. Even standard MEMS’ processes, such as lift-off or sacrificial photoresist releasing, can suffer serious delamination, wherein the parylene separates from other coating or component materials. Because chemical adhesion is impossible, mechanical methods must be used.
For instance, low surface energy materials, such as Kapton (polymide), limit parylene for even mechanical applications, and entirely reject chemical adhesion, causing rapid delamination and peeling of the conformal coating. Even standard MEMS’ processes, such as lift-off or sacrificial photoresist releasing, can suffer serious delamination, wherein the parylene separates from other coating or component materials. Because chemical adhesion is impossible, mechanical methods must be used.
On its own, parylene does not technically adhere to substrate surfaces. It attaches to itself instead, becoming absorbed into the often microscopic pits and cracks typical of some substrate surfaces; once it is so attached, parylene doesn't easily decompose, or otherwise relinquish its adhesion to the substance. However, if parylene is deposited on a perfectly smooth surface, the lack of inherent exterior cracking and pitting offers no imperfections to fill, significantly diminishing coating reliability.
The impossibility of chemical adherence specifies mandatory use of mechanical processes to improve parylene surface adherence. Such is the case when using parylene to conformally coat noble metals.
Points to Consider
Sensors measure specific aspects of data-driven technology. Included are such performance properties as acceleration, fluidity, humidity/temperature, position, pressure or vibration. Sensors collect data and respond with feedback for a multitude of electronic devices utilizing printed circuit boards (PCBs) and related sensitive electronics. They have been successfully adapted for use across a wide range of applications, including aerospace/military, appliance, automotive, communications, consumer, industrial, medical and transportation uses.
PCBs and the larger devices they power often need to function in harsh operating environments. Conformal coatings -- liquid acrylic, epoxy, silicone and urethane resins, and chemical vapor-deposited (CVD) parylene – provide PCBs and similar electronics excellent barrier, dielectric and insulative protection through most performance conditions, sustaining their expected utility. Substrate adhesion is necessary to conformal film reliability; coatings do not work if they delaminate or otherwise disengage from the components they are applied to protect.
Cases emerge where typical application methods cannot guarantee appropriate film adhesion. Surface adhesion promoters – carbonyl or hydroxyl for liquid coatings, silane for parylene – improve the bond between coating materials and substrates. In this respect, evidence shows:
For instance, γ-MPS (γ-methacryloxypropyl trimethoxysilane), a silane coupling agent, promotes adhesion of photolithographically patterned polymer hydrogels to conductometric sensors. Situated on oxidized silicon wafers, sensors subjected to peel-tests of adhesion after immersion in solutions of buffered potassium chloride, with differing pH, as well temperature-changes analysis, showed varied results:
Conformal film’s adhesion is affected by pH sensitivity. Coating failure frequently causes sensor dysfunction. Evidence suggests siloxane bonds between the polymer and the substrate reformed, offering the possibility of sensor regeneration through vacuum oven curing. Additionally, higher-level bonded-energy keeps siloxane bonds from breaking at temperatures of 200°C, while providing additional chemical stability and weatherability. Maintenance of the siloxane bond can promote ongoing film adhesion and prevent sensor failure.
Other evidence exists. Microelectromechanical (MEM) surface acoustic wave (SAW) sensors use modulations of surface acoustic waves to sense multiple physical phenomena within a device’s operating environment. These include force, humidity, mass, pressure, strain and temperature. Water barrier studies of SAW sensors show that introduction of promotion materials improves film adhesion through formation of a chemical- or acid-base type bond for conformal films frequently adapted for MEMS’ uses. Conformal film adhesion promotion lessens the impact of sensor failure mechanisms.
Silane adhesion promoters form unique chemical bond with substrate surface, sufficient to improve conformal coating’s mechanical adhesion. Yet, silane is not effective for all materials or sensor uses. Adhesion promotion options to consider include:
Adhesion failure between conformal film and the substrate is a primary cause of sensor malfunction. As always, much depends upon the sensor’s specific application, its material structure and operating environment. Establishing conformal film adherence solutions for sensors should be an important topic of research in years to come.
Key Factors
Applied as a conformal coating through a unique chemical vapor deposition (CVD) process, parylene provides micron-thin, resilient barrier protection for an exceptional range of electrical assemblies. In comparison to liquid coatings -- acrylic, epoxy, silicon, urethane -- parylene is the coating-of-choice for protecting printed circuits boards (PCBs) and medical devices. It’s films negate the impact of gravity and surface tension during the coating process.
With appropriate pre-treatment, CVD penetrates deep within substrate surfaces, rather than merely attaching to substrate exteriors, as liquid-application coatings do. Ultrathin protection negates operational dysfunction caused by excessive coating mass or viscosity, conditions that can impede assemblies’ performance. Parylene is chemically and biologically inert and stable, generating excellent barrier material to abrasive chemicals, bodily fluids, solvents, liquid water and water vapor. It is also readily adaptable for MEMs/nano technology.
Excellent but Not Flawless
PCBs and like electronics can feature angular surfaces, crevices, exposed internal surfaces, pointed/sharp edges, or similar specialized assembly configurations; they are well-served by parylene’s all-inclusive conformal protection. While these features speak to parylene’s durability, reducing the need for repair and replacement, the coatings are not perfect. the potential for bridging, pinholes, puddling, run-off, sagging, or thin-out common to wet coatings is eliminatedelaminated.
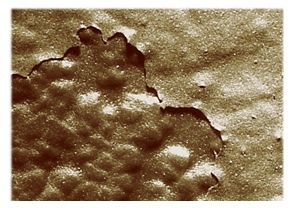 Durable and heat/liquid resistant, the coatings are difficult to rework, and occasionally require repair. Delamination, a lack of adhesion occurring when a conformal coating lifts away from the substrate, exposing the assembly’s workings, is a major parylene failure mechanism, necessitating repair.
Durable and heat/liquid resistant, the coatings are difficult to rework, and occasionally require repair. Delamination, a lack of adhesion occurring when a conformal coating lifts away from the substrate, exposing the assembly’s workings, is a major parylene failure mechanism, necessitating repair.
Delamination and Parylene
Delamination results from faulty adhesion between the coating and substrate, producing a torn, unattached, and non-conformal coating. Separation from localized PCB regions is not necessary; even partial lifting of the parylene coating is sufficient to qualify as delamination. Since appropriate adhesion of conformal coatings is critical to their success, exposing even some fragment of the region to-be-protected negates their purpose and utility.
Delamination is frequently the result of:
Lifting the parylene film from the substrate, delamination leaves an unprotected area that may be subject to further coating peel-back and addition assembly exposure. Care must be taken both prior to and during CVD application procedures to assure subsequent delamination episodes do not occur.
Repairing Delaminated Parylene
Chemically resistant, parylene seldom responds to solvent removal; coated surfaces are resistant to most types of reworking. Parylene films are difficult to:
Specialized techniques -- micro-blast abrasion, laser ablation or plasma etching – successfully remove delaminated parylene coating. Perhaps even more effective is removal by mechanical incision, a costly, labor-intensive, and time-consuming method. Manually enacted, the treatment surface is cut by incisive tools, further lifting already separated film from substrate; the damaged parylene is then removed from the assembly with tweezers or similar implement. Because workers’ unprotected skin can contact the substrate surface during removal procedures, leaving bodily oils, sweat or like residue on the substrate, it is suggested operators wear protective gloves; otherwise, additional cleansing processes will be required, complicating the repair process, while adding to it’s expense.
In most instances, localized, spot removal is all that’s necessary; stripping the entire board is seldom required. Use of a soldering iron to carefully burn through the parylene is recommended in these cases, followed by touch-up with urethane or other liquid coating to tack down the parylene.
Regardless of the repair method, the goal is successfully stripping damaged parylene from the effected surface and restoring it, so assembly function can be maintained. Reworked/removed parylene can frequently be recoated to original specifications.
Prevention of Delamination
Prevention is the best cure. To lower the risks of delamination and the need for subsequent repair, ensure the following prior to CVD:
Post-production and inspection procedures must target the possibility of delamination, to limit its incidence.
Parylene Surface Protection
Parylene conformal coatings provide effective chemical, electrical, moisture, and vapor protection for complex electronic circuit assemblies made to maintain performance throughout demanding operational circumstances. Parylene's effective adherence to the substrate's surface is imperative if its many benefits are to be realized. Mechanical methods of adhesion promotion must be employed to ensure parylene appropriately adheres to substrate surfaces.
Surface Cleanliness
While the objective is achieving the highest levels of uninterrupted performance under the harshest operational conditions, virtually all of parylene's positive qualities are diminished by inadequate adhesion. Dominating the prospect of appropriate adhesion is surface cleanliness. While short-term effects can be minor, this is especially true for longer-term performance; contamination generated by dirty surfaces can lead to severe degradation of affected operating systems, as the parylene coating begins to disengage from the surface.
Unique Application Process Requires Superior Surface Cleaning
Most conformal coatings are applied by either dipping the substrate into a liquid bath of the coating material, or spraying the material directly onto the substrate surface. Parylene's application process is more complex, requiring a vapor-phase, chemical-vacuum deposition process. There is no intermediate liquid phase. The powdered parylene dimer is first converted to gaseous form at the molecular level, through cross-link polymerization, and then applied directly to targeted surfaces as a transparent, polymerized film.
Dirty surfaces prohibit parylene from penetrating the substrate's smallest surface pores. Thus, it is unable to bond with both the material being covered and itself after deposition. These conditions reduce its capacity to generate superior conformal coating.
Thorough cleansing of the surface beforehand is necessary to ensure the absolute conformal adhesion that distinguishes parylene from other substrate coatings. If trace organic elements -- chemicals, dust, oil, waxes, or other residue left during manufacture, handling or transportation -- remain on the surface, the bond between the underlying surface and the applied parylene can be subjected to mechanical stress or thermal cycling. Inorganic compounds similarly disrupt the prospect of surface adhesion.
Inspection to Detect and Identify Potential Contaminants
These conditions can eventually nullify the coating's beneficial attributes, as delamination occurs, eventually exposing the surface. Electrically conductive, non-organic deposits beneath the parylene can leak current during usage of electronic components, distressing their functionality. Costly cleaning issues can emerge if thorough inspection of surfaces is overlooked at any stage during the production/coating process. Inappropriate inspection for contaminants may compel reworking covered surfaces and similar production delays. Recommended methods of inspecting surfaces include:
The objective in all cases is determining the optimal solvent and cleaning system suitable for eradicating the contaminant.
After precise identification of surface contaminants, numerous cleaning agents can be effectively applied. Using nonhazardous cleaning materials is advised in all cases. Soluble contaminants generally respond to regular detergent cleaning. Multi-faceted, solvent-strength solutions are necessary to assure surfaces are free from less soluble substances. Safe, biodegradable solvents available for these purposes include deionized water, isopropyl, and methyl ethyl, among others developed in recent years. Depending on the contaminant substances and the requirements for acceptable levels of cleaning, such methods as solvent immersion, spray, tumbling, or vapor-degreasing can be applied; in some cases even cleaning by hand may be the best approach to assure surfaces are clear for parylene adhesion.
A174 Silane Adhesion Promoter
Production and material considerations often recommend the use of A174 silane adhesion promoter for optimal substrate surface-cleansing and subsequent parylene adhesion. Substrates responding well to treatment with A174 silane prior to implementation of parylene coating processes include elastomer, glass, metal, paper and plastic, among many other materials. Application of A174 silane after the masking-operation generates consistently reliable results, either through manual-spray, soaking, or vapor-phase processing. Silane's chemical-bonding with the substrate surface promotes resolute parylene adhesion.
Despite its value as a promoter of enhanced parylene adhesion, A174 is not effective for all materials or uses. Researchers seek additional cleansing/adherence agents to improve parylene's conformal utility for these purposes. For instance, parylene delamination problems attendant to medical devices have been reduced through application of plasma surface-treatment methods. Development of additional cleansing/adherence solutions and strategies will be a primary focus of the parylene community through the near future.