Parylene's deposition process is unique among conformal coatings. Unlike others that start as a liquid, get deposited and dry, it starts as a solid. Parylene coating equipment turns it into a vapor, where it then deposits onto the substrate. This unique four-step method poses some challenges but also brings real advantages.
How it works
Sublimation
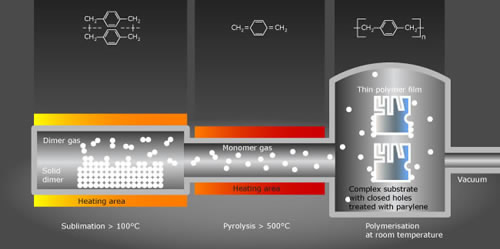
In the first step of the process, the powdery raw material, called dimer, is heated. As the temperature approaches 150 degrees Celsius, the dimer sublimates into a gas without passing through a liquid state. At this point, the dimeric gas is ready to pass into the next stage of the deposition process.
Pyrolysis
The dimer gas actually consists of two parylene molecules, bonded together. In the pyrolysis stage, the gas is further heated in a furnace to temperatures as high as 700 degrees Celsius. The heat splits the molecules apart -- creating a monomer. That monomeric vapor is what becomes the parylene conformal coating when it reaches the item to be coated in the deposition stage.
Deposition and Polymerization
The container holding the items to be coated is depressurized to create a vacuum. That vacuum sucks the parylene gas out of the pyrolysis chamber and into the deposition chamber. In that chamber, which is maintained at normal room temperature, the parylene vapor turns into a polymer, coating everything that it touches. During the deposition process, the parylene gas cools without having any meaningful heating effect beyond a few degrees on the items being coated.
Waste Collection
The vacuum pump that pulls the parylene gas through the deposition chamber also plays an important role in disposing of waste vapor. As the gas exits the chamber, the pump pulls it though a cold trap. That trap, which is usually cooled to extreme subzero temperatures, causes the parylene to solidify so that it can be disposed of.
Challenges in the Process
Adhesion is generally one of the largest problems with parylene, especially when trying to coat noble metals. This is often addressed by applying silane A-174 or other adhesion promoters prior to or during the deposition process.
In addition, because of parylene's unique method of deposition, quantities can be limited. Put simply, you can only coat as many items as will fit in the deposition system's coating chamber. While these systems can be relatively large, there are still practical limits to how many items you can coat at one time.
Furthermore, while parylene coatings are typically thin, they also deposit relatively slowly. The fastest-depositing variant of parylene -- Parylene C -- typically deposits at a rate of 0.2 mils or 5 microns per hour. This means that a 75 micron coating would take approximately 15 hours. Parylene N and D deposit more slowly.
The Power of Vapor
Even given some of the drawbacks inherent in the parylene deposition process, using a vapor-phase coating has significant advantages. For example, because the vapor is essentially dry and because the process occurs at room temperature, even very sensitive items can be safely coated without any damage.
Parylene vapor can go anywhere that air can. This means that it coats under parts, inside small crevices and can even coat inside semi-sealed areas. Its thinness means that it can coat without forming bridges in tight areas as well.
A parylene coating is also truly conformal, since it does not run or drip on the surface the way that some liquid coatings can. Since it deposits on the entire surface at the same time, it also creates an extremely even coating of the same thickness, everywhere. This characteristic is one of the reasons that it is also typically the most pinhole free of all of the conformal coating compounds.
Ultimately, the parylene deposition process is an important part of what makes parylene such a desirable coating choice. While its vapor-phase deposition can be time-consuming and expensive relative to other coatings and their more traditional application methods, its results are also unparalleled for many applications.
How temperature affects Parylene
Parylene (XY) conformal coatings are applied to substrate materials through a specialized chemical vapor deposition (CVD) process that completely eliminates the liquid phase of wet coatings. No initiators or catalysts are involved in CVD polymerization, which synthesizes truly conformal protective film in-process. This is in stark contrast to wet coating materials such as acrylic, epoxy, silicone and urethane, which are synthesized prior to application via, brush, dip or spray methods. Wet during application, liquid-coated substrates requiring further drying and curing.
With parylene CVD deposition, a gaseous monomer uniformly encapsulates all exposed substrate surfaces; no curing is necessary. Temperature levels influence outcomes of CVD throughout the process, from sublimation through pyrolysis and cold trap procedures.

Sublimation – Dimer Transformation
In contrast to liquid coating materials, parylene initiates CVD as a solid, stable powdered crystalline dimer di-p-xylylene. CVD’s first phase -- sublimation – begins with positioning dimer in an aluminum foil cup – the “boat” -- situated at opposite end of the deposition chamber, where actual coating will ultimately take place, Final coating thickness is determined by the volume of dimer placed in the boat.
CVD processing begins with radiant heater cycling within the unit.
Regarding process temperature, dimer is heated to levels between 120° - 150° Centigrade (C) under vacuum conditions, transforming the substance into a vapor. A pressure safety interlock cycles the radiant heater/vaporizer on-and-off according to coating project requirements, to regulate safe operating performance. As dimer changes from solid-to-vapor, its molecules move down the tube because of the reduced pressure at the opposite end.
Parylene Pyrolysis
Pyrolysis is essentially decomposition of a substance, in this case solid parylene dimer, brought about by exposure to high temperatures. Consisting of two parylene molecules, the dimer undergoes this transition at temperatures ranging between 650° - 700° C. This level of heat causes the molecules to split apart forming the reactive, vaporous monomer p-xylylene. This resulting monomeric vapor becomes the parylene conformal coating when it reaches the item to-be-coated in the deposition stage.
Cleaved into divalent radical monomers by these high temperatures, monomer molecules:
Averaging about 680° C, this temperature initiates the polymerization described above, reliant on physisorption -- a function of deposition pressure and temperature. Because of surface energy at the interface of vaporous parylene and the substrate, the gaseous molecules adhere to its surface, depositing a conformal film. Physisorption’s kinetic properties are stronger at lower temperatures than higher.
Cold Trap Processes
Drawn one molecule at-a-time onto the selected substrate, monomeric XY gas reaches the final deposition phase, in the cold trap. Temperatures are cooled well below zero (0º C), between -90º and -120º C. This process allows for:
At temperatures below its freezing point, the monomer condenses as a crystalline solid. The freezing point of Parylene N registers at approximately -73° C after thermal measurement; for Parylene C, freezing point is higher, closer to -65° C.
Threshold Temperature
Common to all parylenes, the threshold temperature is that thermal level essentially negating film deposition. Also known as the ceiling temperature, further coating deposition is negligible. Physisorption diminishes incrementally as one nears the threshold temperature, slowing deposition until none occurs when ceiling is reached. Once physisorption begins, the p-xylylene intermediate needs to react with itself to assure polymerization.
Parylene threshold temperatures vary according to parylene type, and each type’s molecular weight. The greater the molecular weight, the higher the threshold temperature; consult the following table:
| Parylene Type | Threshold Temperature |
|---|---|
| C | 90°C |
| D | 135°C |
| N | 40°C |
| AF-4 | 30°C - 35°C |
Additional Thermal Data
Combining high thermal stability with a low dielectric constant, CVD-generated parylene films also provide sustained substrate adhesion, characterized by minimal moisture absorption. Further beneficial thermal properties of XY protective coatings include reliable performance through an exceptional range of temperatures. Depending on the parylene type, XY can function at temperatures as low as -271º C, or as great as 450º C, representing total sustained operation within a thermal span of 721º C.
The following table provides additional significant temperatures pertinent to using parylenes C, D and N.
| Properties | Parylene C | Parylene D | Parylene N |
|---|---|---|---|
| Melting point | 290°C | 380°C | 420°C |
| Continuous service
temperature (O2/inert environments, °C ) |
80°C - 230°C | -- | 60°C - 220°C |
| Short-term service
temperature (per 1,000 hours use, O2/inert environments °C) |
110°C - 320°C | -- | 85°C - 265°C |
| T5 point, °C (modulus = 690 MPa) | 125°C | 125°C | 160°C |
| T4 point, °C (modulus = 70 MPa) |
240°C | 240°C | 300°C |
| Thermal conductivity, 25°C | 2.0°C | -- | 3.0°C |
| Specific heat, 25°C | 0.17°C | -- | 0.20°C |
Key planning concepts
The parylenes consist of a range of para-xylylene polymers whose desirable physical and electrical properties support expansive utilization as conformal coatings for electronic and medical devices Parylene films are applied to substrates via a chemical vapor deposition (CVD) process, which deposits monomeric parylene vapor homogeneously and deeply into the surface of printed circuit boards (PCBs) and related assemblies/components.
In comparison to liquid conformal coatings – acrylic, epoxy, silicone or urethane -- parylene offers highly desirable physical and electrical properties, performing where wet coatings might be compromised or insufficient. Among parylene’s many advantages are:
Despite these advantages, the application of parylene film to PCBs is complicated by several failure mechanisms that require adroit management to assure successful implementation of the CVD procedure. One of the most important is supervision of the batch process, wherein designated consignments of PCBs and other products undergo parylene application in the deposition chamber.
Batch Processing as a Production System
Typically, batch process production manufactures goods in sequential stages, that are in line but disconnected for performances at different workstations. Each lot of products – in this case PCBs, medical devices or other related items – comprises a batch coated by parylene CVD. Normally, batch process workstations are situated closeby in a manufacturing facility; for parylene coating, a vacuum chamber is used for each coating batch, with processes following a progression that leads to a final stage, deposition of the coating.
Implemented in a closed-system vacuum subjected to persistent negative pressure, the Parylene process integrates the following steps as part of the batch coating process:
Chambers are typically small, which can limit batch size. The flexibility of batch processing supports just-in-time (JIT) manufacturing. Lean JIT batch-runs respond specifically to customer demands, largely eliminating outsized product inventories, while reducing waste in manufacturing/warehousing procedures. With parylene dimer materials costing between $200.00 - $10,000.00 per pound, and the need for worker interaction throughout production, a major consideration in managing CVD is the minimization of wasted resources and effort.
Aspects of Parylene Batch Processing
As described above, parylene batch coating procedures differ appreciably from conventional production line operations. Composed of a succession of substrate pre-treatment stages, coating processes are documented through a formal control plan that specifies batch size; methodical logistics for implementing the batch coating require controlled staging to generate both production efficiency and cost-effective coating operations.
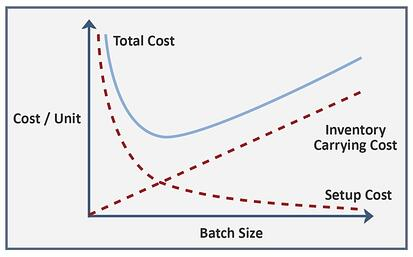
The batch coating process requires structured handling and defined time periods. Regarding chamber load, if the same parylene type is used and the coating specifications are equivalent, the actual amount of time the product to-be-coated undergoes parylene deposition varies very little, when comparing small to larger batches. In general, time-consuming CVD is confined to small-batch production, which can vary between 8 – 24 hours, despite smaller batch-size.
Thus, a greater quantity of product in the deposition chamber for coating generally leads to a more economical batch coating process. However, CVD technology has built-in limitations that negate further reduction of chamber time, regardless of batch size. Therefore, real-time coordination between production and quality control processes must be rigorously synchronized to minimize wasted time and resources, while maintaining client specifications, consistently between orders. To this end, the parylene CVD process is effective because of:
Yet, these factors do not much diminish the generally limited throughput of parylene batch processing. Time-consuming CVD reduces operational efficiency, particularly with small-batch production dominating most coating assignments. Deposition chambers are somewhat costly and tend to be physically small, reducing total quantity of product coated during any single coating session. Turnaround-time required to complete coating assignments places further constraints on batch-size, a condition that cannot be overstepped since processes must be fully completed to assure successful coating application. Unused chamber volume reduces cost efficiency, so closely matching the batch size to the chamber capacity contributes to cost effectiveness. Diamond maintains both larger and small chambered coating machines to better optimize customer/company expense on coating assignments.
Key principles
Parylene deposition is a complicated process that needs to be effectively monitored to ensure its superior levels of protection and performance. However, inappropriately applied parylene can sometimes have an adverse effect on the functionality, integrity, and performance of an assembly or component. Masking selected regions of a substrate is an essential part of the parylene coating process.
Examples of Potential Product Failure
Parylene conformal coating does an incomparable job of protecting product substrates from harsh, unpredictable operating environments of all kinds. However, while most of parylene's key properties -- barrier strength, dielectric, flexibility, lubricity, etc. -- are exceptionally useful to a part or assembly, much depends on where the conformal coating is applied. Because parylene coatings are so effective, covering inappropriate areas of a component can have a negative impact on its overall performance, defeating the basic purpose for parylene application.
In this respect:
In the case of printed circuit boards (PCBs) and similar electrical devices, parylene's superior dielectric qualities are detrimental, precisely because they conduct electricity poorly. Masking ensures electrical contact points are uncovered and operable, allowing them to fulfill their function. Parylene covering of moving parts can not only retard their motion, and thus impede their functionality, but will also damage the coating itself, diminishing the overall integrity of the parylene coating throughout the component.
Masking provides the best solution yet devised for these circumstances, which are common to many of parylene's product uses. By ensuring the circuit board, contact point or moving part remains uncoated, while the remainder of the product is effectively and conformally covered, masking resolves these issues; the many advantages of parylene are accessed, without suppressing the product's ability to function as designed.
The Parylene Masking Process
While very necessary, the parylene masking process is also costly and time-consuming. Several steps encompass the masking process and ensure proper implementation:
Do It Right
You'll want to make sure the masking process is properly implemented. Not only is this necessary to preserve the product's integrity, guaranteeing it works as designed, but also because of parylene's superiority as a conformal coating. Very simply, once bonded to a substrate, parylene is exceptionally difficult to remove. If masking is unsuccessful, further time and expense will be wasted removing parylene from the product's sensitive regions, potentially damaging the conformal coating throughout the product. The expensive, time-consuming processes of both masking and coating will have to be repeated to assure product functionality and protection.
Precautions to consider
Application of parylene’s xylylene monomer employs a chemical vapor deposition (CVD) process implemented under a vacuum. Unlike wet coating application methods – brushing, dipping, spraying, etc. – parylene CVD is not line-of-sight. Because the vaporous monomer envelopes all sides of the assembly being coated, appropriate process control allows vacuum deposition of an entirely conformal coating, one that penetrates deep into any crevices, rivulets, or sharp edges and points that exist on the assembly’s surface. The resultant parylene film is insulating, ultra-thin, and pinhole-free, exhibiting superior protective barrier qualities and very low moisture permeability.
Among many others, these properties distinguish parylene as a conformal coating, when compared to liquid coatings such as acrylic, epoxy, silicone and urethane. Exceptionally stable biologically, chemically and electrically, parylene’s versatility recommends its usage for a wide range of products and purposes, including aeronautic, automotive, consumer, electronic, medical and military, encompassing MEMs- and nanotechnologies. Nevertheless, it is always necessary to establish parylene’s capacity for prolonged, safe-use for whatever coating application it is directed to.
Safe-use of Parylene in Storage and Manufacture
A white, granular (powdered) solid with a distinctive, organic odor, parylene’s health hazards are limited, but need to be monitored. While its reactivity hazards are negligible, parylene can be dangerous to workers due to over-exposure, which causes potential irritation to the handler’s eyes, skin or mucous membranes.
A minor fire hazard also exists. At higher temperatures, parylene may ignite and burn, generating potentially irritating vapors and toxic gases, like carbon dioxide, carbon monoxide and hydrogen chloride, due to thermal decomposition from excessive temperature. Furthermore, detectable quantities of dioxins, furans and a variety of other substances may result under certain conditions.
While these problems are undoubtedly serious, they can be easily controlled with careful worksite monitoring. Parylene infection targets the respiratory system, skin, and eyes at the ACUTE level of toxicity; skin problems can also reach CHRONIC levels if prolonged exposure is unchecked.
Inhalation of dusts and contact with skin and eyes represent the most likely conduits of occupational overexposure. Workers’ respiratory systems, particularly the nose and throat, are most significantly infected by inhalation of parylene-derived dust or particulates. Severe coughing, sneezing, or sore throat are the most evident symptoms of such parylene over-exposure in the workplace. The symptoms are typically alleviated when the afflicted individual is separated from exposure and given access to fresh air. If this is insufficient to generate recovery, artificial respiration should be instituted to support improvement of the victim’s vital functions.
Parylene that comes into contact with skin may instigate mild-to-moderate irritation, particularly after prolonged exposure. Dermatitis (reddened, dried skin) may develop if the worker’s exposure to parylene is especially intensive and protracted. While no parylene components are known to be absorbed through the skin, contact with eyes can result in irritation, reddening, and pain. After skin contamination, running water should be used to sanitize the affected area. Flushing should continue no fewer than 15 minutes; additional cleansing is suggested if any sign of skin irritation develops. Immediate medical attention needs to be provided workers victimized by the development of any antagonistic exposure symptoms. Eye exposure also requires liquid decontamination with gently running water, again uninterrupted for a minimum of 15 minutes. Medical attention must be provided as necessary.
Neither ingestion nor injection of parylene are major conduits of worker overexposure, with incidents of swallowing (etc.) far outnumbering those of accidental injection (being jabbed by a parylene-contaminated object). Inflammation of the mouth, throat, esophagus, and other digestive tissues can occur upon contact if parylene is swallowed. Excessive ingestion can incite nausea, stomach-ache, vomiting, and related gastric disorders. Contacting a physician or poison control center is recommended if parylene is swallowed. The victim’s mouth should be rinsed thoroughly with water. Vomiting should NOT be induced, although it should be assisted by leaning the victim forward if it occurs naturally. However, those unable to swallow, having convulsions or rendered unconscious should receive NO dilutents, and be treated by a physician immediately. Injection causes a wound at the site of the puncture, with attendant irritation, swelling and pain.
In all cases, victims should be taken for medical attention if they feel unwell or if adverse effects occur. Such pre-existing conditions as:
may be aggravated by over-exposure to parylene. To prevent these occurrences, worksites are advised to ensure reliable ventilation throughout manufacturing areas. Compliance with germane regulations -- local, state, federal and international – governing the use of parylene is mandatory. Where excessive exposure occurs, covering or outright removal of contaminated materials is recommended to avoid subjecting rescuers to the dangerous conditions. Emergency responders are strongly advised to wear protective equipment best-suited to the particular circumstances of the response situation, in those rare occurrences where parylene handling becomes unsafe.
Consumer Safety of Parylene-coated Products
One category of products where parylene coatings are an integral to the product is implantable medical instruments, such as blood pressure sensors and cardiac-assist devices like pacemakers. Other medical devices benefitting from parylene film protection include:
Consumer safety is generated because the devices themselves maintain operation in conditions where reliable functionality may be necessary to sustain the patient’s life.
Parylene:
Exceptionally stable biologically, chemically and electronically, parylene provides an effective electrical insulator that will not degrade in the presence of electrical current. It resists solvents and is insoluble at temperatures to 150°C. These factors generate reliable consumer safety under most performance circumstances, for the majority of parylene’s uses. However, repeated cyclic stressing MAY generate stress cracking and pinholing of parylene coatings for implants after prolonged use (2+ years), a situation requiring accurate monitoring.
Biocompatible, parylene possesses no pigment or similar properties that might:
Moreover, parylene’s ultra-thin, dielectric coatings can usefully be applied to MEMS or nano medical technologies, designed for use in less accessible, more-constricted bodily regions, offering medical service otherwise unavailable to patients. Parylene’s durable biocompatibility has no negative impact on bodily functions; implants enhance organ/tissue performance, and ensure the device's acceptance in these critical, difficult-to-reach regions of the body. Superior medical product service includes better lubrication along devices’ surfaces, improving patient comfort, further supporting consumer safety. In all, parylene-protected implants assure the lowest level of negative and potentially infectious surface-to-tissue interaction of any currently available conformal coating.
In addition to its utility coating medical devices, parylene's reliable combination of adhesion, electrical conductivity, durability, and flexibility augments consumer safety for other products:
Conclusion
In all, parylene coatings are chosen for a wide range of both consumer and specialized products because they generate consistently reliable protection, shielding components and exteriors without diminishing their functionality. In this way they add value and consumer safety.